

Make smart decision with smart alternative!
OpenTex® Non-Resorbable PTFE Membrane is a pure medical-grade polytetrafluoroethylene (PTFE) sheet with inert biological features and predictable barrier effect. Due to the smooth surface and small pore size, OpenTex® PTFE Membrane resists the incorporation of bacteria into its structure and eases the removal of the membrane.
EU-HP-OPNN-0
MAIN FEATURES

Non-Resorbable
![]() 100% medical grade PTFE
membrane.
100% medical grade PTFE
membrane.

100% medical grade PTFE membrane.
![]() Biologically inert and chemically non-reactive.
Biologically inert and chemically non-reactive.

Biologically inert and chemically non-reactive.
![]() Healing procedure is not interfered with membrane absorption.
Healing procedure is not interfered with membrane absorption.

Healing procedure is not interfered with membrane absorption.

Non-Resorbable
![]() Promote the gingival tissue attachment.
Promote the gingival tissue attachment.

Promote the gingival tissue attachment.
![]() Enhances ease in the interstitial fluid circulation.
Enhances ease in the interstitial fluid circulation.

Enhances ease in the interstitial fluid circulation.
![]() Resist the bacteria infection and fibroblast cells migration.
Resist the bacteria infection and fibroblast cells migration.

Resist the bacteria infection and fibroblast cells migration.

MINIMALLY INVASIVE
![]() Rapid recovery of soft issue.
Rapid recovery of soft issue.

Rapid recovery of soft issue.
![]() Primary Closure is not necessary.
Primary Closure is not necessary.

Primary Closure is not necessary.
![]() Virtually imprevious to bacteria.
Virtually imprevious to bacteria.

Virtually imprevious to bacteria.
![]() Minimum flap reflection or dissection.
Minimum flap reflection or dissection.

Minimum flap reflection or dissection.
![]() Safe from bacteria infection, even in the event of the exposure.
Safe from bacteria infection, even in the event of the exposure.

Safe from bacteria infection, even in the event of the exposure.

Withstands Exposure
![]() Protect the tissue regenaration site.
Protect the tissue regenaration site.

Protect the tissue regenaration site.
![]() Regenerated underlying tissue can be evaluated.
Regenerated underlying tissue can be evaluated.

Regenerated underlying tissue can be evaluated.
![]() Provide a proper environment for the growth of blood vessel and osteogenic cells.
Provide a proper environment for the growth of blood vessel and osteogenic cells.

Provide a proper environment for the growth of blood vessel and osteogenic cells.
OpenTex®
BENEFITS
BENEFITS
Precisions slim cut trianglar needle for small penetration area and smooth suturing.
Minimize damage to surrounding soft tissue.
OpenTex®
STRENGHTS
STRENGHTS
Stability
Non-resorbable PTFE Membrane offers enough healing time to bone regenerative process.
Biologically Inert
PTFE is soft tissue friendly so it is ideal material as a barrier for bone regenerative process.
OpenTex®
CHARACTERISTICS
CHARACTERISTICS
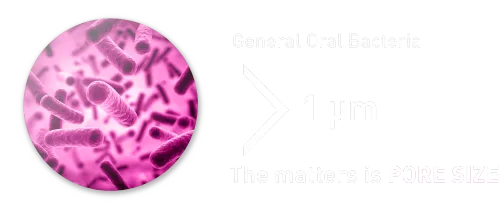
Impervious to Bacteria
Most of Oral Bacteria is larger than 1um. OpenTen® is micro-porous material that has the pore size small enough to prevent bacterial infiltration.
Biocompatible, OpenTex® facilitates cell adhesion on the surfaces.
Test performed shows that the surface of OpenTex® is not toxic causing cells to adhere well on the surface.
OpenTex®
BENEFITS
BENEFITS
Precisions slim cut trianglar needle for small penetration area and smooth suturing.
Minimize damage to surrounding soft tissue.
OpenTex®
STRENGHTS
STRENGHTS
Stability
Non-resorbable PTFE Membrane offers enough healing time to bone regenerative process.
Biologically Inert
PTFE is soft tissue friendly so it is ideal material as a barrier for bone regenerative process.
Withstands to exposure
PTFE Membrane withstands to exposure since it is impervious to bacteria due to their barrier function.
OpenTex®
CHARACTERISTICS
CHARACTERISTICS
Impervious to Bacteria
Most of Oral Bacteria is larger than 1um. OpenTen® is micro-porous material that has the pore size small enough to prevent bacterial infiltration.
Biocompatible, OpenTex® facilitates cell adhesion on the surfaces.
Test performed shows that the surface of OpenTex® is not toxic causing cells to adhere well on the surface.

GBR (GUIDED BONE REGENERATION)

Simultaneous use of GBR membrane and implants.
![]() Simultaneous use of GBR membrane and implants.
Simultaneous use of GBR membrane and implants.

Augmentation around implant placed in immediate extraction sites or delayed extraction sockets.
![]() Augmentation around implant placed in immediate extraction sites or delayed extraction sockets.
Augmentation around implant placed in immediate extraction sites or delayed extraction sockets.

Filling of bone defects after root resection, removal of cysts and removal of retained teeth.
![]() Filling of bone defects after root resection, removal of cysts and removal of retained teeth.
Filling of bone defects after root resection, removal of cysts and removal of retained teeth.
GTR (GUIDED TISSUE REGENERATION)
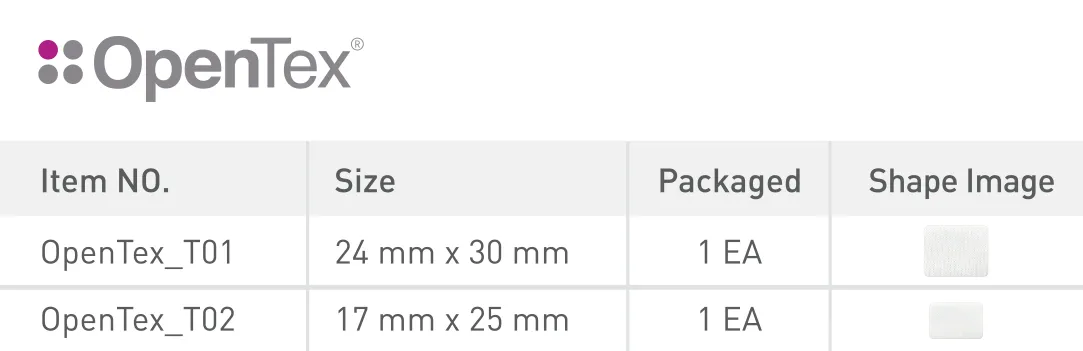
Bibliographical references
Implantology 2018; 22(1): 2-11
Clinical Evaluation of Vertical Ridge Augmentation Using Titanium Reinforced PTFE membrane

Non-resorbable PTFE membrane.
Meet Purgo Solutions























